According to medical professionals in Michigan, magnetic resonance imaging (MRI) may be used to spot fungal infections caused by the tainted steroid injections that led to last year’s deadly meningitis outbreak. The researchers published their findings in the Journal of the American Medical Association on June 19, as a result of tests that were conducted to help deal with the fungal meningitis outbreak that has killed nearly 60 people and sickened more than 700 more nationwide. The study authors from St. Joseph Mercy Hospital say that the technique could be used to detect fungal infections in patients before they exhibit any symptoms, and in patients who remain asymptomatic for a long period of time after receiving the injections. If you received a steroid injection in Tennessee and you believe you may have been exposed to fungal meningitis or another serious illness, contact our reputable attorneys at Michael D. Ponce & Associates for legal help.
Dangers of Contaminated Medications
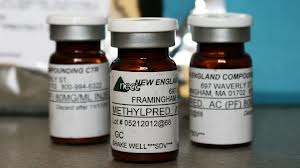
The medication at the center of the fungal meningitis crisis was a tainted epidural steroid injection mixed by a compounding pharmacy in Massachusetts. The New England Compounding Center (NECC), which was shut down in the aftermath of the outbreak, recalled 17,676 vials of the methylprednisolone acetate injections after discovering that they may have been contaminated with a mold called aspergillus. Unfortunately, prior to the recall, the tainted injections had already been distributed to 75 health care clinics in 23 states in the U.S., including Tennessee. As part of this latest study, researchers screened 172 patients who had received steroid injections mixed by NECC, finding abnormal MRI results in 36 of them. All but one of the 36 were later diagnosed with confirmed or probable fungal spinal or paraspinal infections requiring anti-fungal treatment.
Contact Our Attorneys to File a Claim
Although the fungal meningitis outbreak is finally winding down, there were still new cases being reported as recently as April, despite estimates by federal regulators that reported cases would taper off at the end of last year. “Screening MRI led to identification in patients who had minimal or no symptoms of spinal or paraspinal infection and allowed early initiation of medical and surgical treatment,” the researchers noted in their report. These findings are particularly important, considering less than two weeks ago, the U.S. Food and Drug Administration (FDA) issued another recall for all sterile products distributed by Main Street Pharmacy in Tennessee, after federal investigators found vials of steroids contaminated with fungus and bacteria. If you believe you have been adversely affected by a contaminated steroid injection in Tennessee, our experienced lawyers at Michael D. Ponce & Associates can help.
Source: http://jama.jamanetwork.com/article.aspx?articleid=1697964